Influenza News
Influenza remains a major health burden despite the use of vaccines.
Honolulu, Hawaii - A recent study published in the New England Journal of Medicine found that influenza remains a major health burden despite the use of licensed vaccines. Nucleoside-modified messenger RNA (modRNA) influenza vaccines have shown promising immunogenicity against influenza and an acceptable safety profile in a phase 1–2 trial. In this phase 3 trial, researchers randomly assigned healthy adults between the ages of 18 and 64 years to receive either a quadrivalent modRNA influenza vaccine (modRNA group) or a licensed inactivated quadrivalent influenza vaccine (control group) during the 2022–2023 influenza season in the United States, South Africa, and the Philippines. The primary end point was relative efficacy, defined by the reduction in the percentage of participants with laboratory-confirmed influenza associated with influenza-like illness at least 14 days after vaccination with the modRNA vaccine, as compared with the control vaccine, and analyzed for noninferiority and superiority. Immunogenicity was evaluated by means of a hemagglutination inhibition (HAI) assay. They assessed reactogenicity within 7 days after vaccination, adverse events through 1 month, and serious adverse events through 6 months. They assessed vaccine efficacy, immunogenicity, and safety in the modRNA group. A total of 18,476 participants underwent randomization: 9225 were assigned to receive the modRNA vaccine and 9251 to receive the control vaccine. The relative efficacy of the modRNA vaccine as compared with the control vaccine against influenza-like illness was 34.5% (95% confidence interval [CI], 7.4 to 53.9) on the basis of 57 cases in the modRNA group and 87 cases in the control group, a finding that met the criteria for both noninferiority and superiority. Cases of influenza-like illness were caused by A/H3N2 and A/H1N1 strains but almost no B strains. The noninferiority of the antibody response on HAI assay was shown for influenza A strains but not for B strains. Primarily mild or moderate reactogenicity was observed in both vaccine groups but was reported more frequently in the modRNA group (overall local reactions, 70.1% vs. 43.1%; overall systemic events, 65.8% vs. 48.7%). Fever occurred in 5.6% of the participants in the modRNA group and in 1.7% of those in the control group. Adverse event profiles were similar in the two groups. The researchers concluded that the modRNA vaccine had statistically superior efficacy over the control vaccine, with greater immune responses to A/H3N2 and A/H1N1 strains but was associated with more reactogenicity events.
For more information: https://www.nejm.org/doi/full/10.1056/NEJMoa2416779
Canada needs a national vaccination registry
Hamilton, ON – This excerpt is taken from an opinion piece, written by Dawn Bowdish*, that appeared in a recent edition of Maclean’s magazine. “Measles is the most contagious virus on earth. It creates scenarios that could have been ripped from science fiction—for example, a woman walks into a room where someone coughed two hours earlier and catches a virus that could permanently damage her brain. As an immunologist, scientist and professor of medicine, my life’s work is understanding how vaccinations can prevent situations exactly like that one and protect health in the long term. You could say I’m the opposite of an anti-vaxxer. After measles broke out in New Brunswick last October, I was hit with a sinking feeling. Its rapid spread either meant that Canada’s health-care system didn’t have the resources it needed to control it, or our MMR vaccine uptake wasn’t as high as it should’ve been—or both. Uptake rates for most childhood vaccinations have been dropping in Canada for years; in the case of MMR, they fall way short of the threshold required for herd immunity. People have blamed the misinformation that ran wild during the pandemic—say, that vaccines are linked to fertility issues—but that’s only part of the problem. In many provinces, kids are still required to be vaccinated for a range of diseases, including measles, before they’re allowed to attend public school. But enforcement for showing proof of vaccination has dwindled. Most provinces also recently added “conscience exemption” to the list of non-medical reasons to opt out of vaxxes—no doctor sign-off required. One of the biggest barriers to vaccinations, however, is tracking. Thanks to the family doctor shortage, many families simply miss their jab appointments without realizing it; they don’t have a medical office to remind them. They struggle to independently navigate Canada’s complex vaccine schedule, which mandates 12 different shots for kids in the first year and a half of their life. It’s no coincidence that we now rank in the top 10 countries globally for measles outbreaks, alongside countries like Yemen and Afghanistan. We’re the only G7 country without a national vaccination registry—and we need one stat.”
For more information: https://tinyurl.com/4ywxuc4c
Dawn Bowdish is a professor of medicine at McMaster University and the executive director of the Firestone Institute for Respiratory Health.
High-dose flu vaccine demonstrates superior protection for older adults against hospitalizations vs standard-dose
Paris, France - Compared to standard-dose influenza vaccines, Efluelda/Fluzone High-Dose demonstrated a reduction in laboratory-confirmed influenza hospitalizations by an additional 31.9% (95% CI, 19.7 to 42.2; p<0.001) in adults 65 years and over. Results come from FLUNITY-HD, the largest influenza vaccine effectiveness study of individually randomized older adults, involving nearly half a million participants across several seasons and two geographic areas. Efluelda/Fluzone High-Dose also provided superior protection, compared to standard-dose influenza vaccines*, against hospitalizations due to pneumonia or influenza, hospitalizations caused by cardio-respiratory events, and all-cause hospitalizations in older adults. Efluelda/Fluzone High-Dose is the first vaccine to demonstrate superior protection against both influenza infection and hospitalizations, compared to standard-dose influenza vaccines, in individually randomized studies in adults 65 years and over. New data from the FLUNITY-HD study, published on October 17 in The Lancet, demonstrated that Sanofi's Efluelda (known as Fluzone High-Dose in North America) significantly reduced the risk of hospitalization in adults 65 years and older compared to standard-dose influenza vaccines. FLUNITY-HD is the largest study of its kind, conducted across multiple seasons.FLUNITY-HD provides robust evidence that the high-dose influenza vaccine offers superior protection compared to standard-dose. Beyond clinical evidence, these findings point to potential public health and societal benefits. “Adults 65 and older represent up to 70% of flu hospitalizations. The FLUNITY-HD data confirm that our high-dose flu vaccine provides superior protection against hospitalizations compared to standard-dose vaccines in older adults,” said Bogdana Coudsy, MD, Global Head of Medical, Sanofi, Vaccines. “For every 515 older adults who receive our high-dose flu vaccine instead of standard-dose vaccines, one all-cause hospitalization is prevented. This can mean a lot, especially for vulnerable seniors, decreasing the burden on their quality of life and helping them to maintain their autonomy for longer. Additionally, preventing influenza hospitalizations may bring societal benefits such as lower healthcare costs, less pressure on medical systems, and reduced burden on caregivers.”
To review the FLUNITY-HD study: https://www.thelancet.com/article/S0140-6736(25)01742-8/abstract
Flu hospitalizations hit record highs last season
Atlanta, GA - Influenza-associated hospitalizations reached record overall levels and peak-weekly marks during the 2024-2025 season, surpassing all rates over the prior 14 seasons, according to an analysis of nationwide surveillance data. The 2024-2025 season's cumulative flu-related hospitalization rates were 1.8 times to 2.8 times greater across all age groups than the median cumulative rates in the seasons since 2010-2011 (127.1 vs 62 per 100,000), according to researchers led by Alissa O'Halloran, MSPH, of the CDC in Atlanta. Last season's weekly flu hospitalization rate peaked in early February 2025, exceeding the previous weekly high mark set in 2017-2018 (13.5 vs 10.2 per 100,000), they noted in the Morbidity and Mortality Weekly Report. "Given similar prevalences of severe disease indicators this season compared with past seasons, the 2024-25 season's severity was likely driven by higher incidence rather than atypical clinical severity," the researchers said. "High rates observed during the 2024-25 season could have been driven by recent lower influenza vaccination coverage in the general population, as well as virus characteristics." The CDC recommends annual flu vaccination for everyone 6 months and older without contraindications. The researchers used data from the FluSurv-NET surveillance system, which includes information on flu-associated hospitalizations in patients of all ages in about 300 hospitals in 14 states. The system covers 9% of the U.S. population. They analyzed hospitalization rates from the 2010-2011 season (range 8.7 to 102.9 per 100,000) through last season. The analysis did not include the 2020-2021 flu season, when there weren't enough flu-related hospitalizations to make rate estimates. Flu hospitalization rates in the 2024-2025 season were greatest in adults 75 and older, at 598.8 per 100,000 (second only to the rate of 726.5 in 2017-2018), and lowest in children and teens ages 5 to 17 years (39.3 per 100,000). Percentages of underlying medical conditions last season matched median numbers from prior seasons. Among children ages 0 to 4 years and 5 to 17 years, asthma was the most common underlying condition (14% and 35.9%, respectively). Among adults ages 18 to 49, obesity was the most common such condition (43.9%), while chronic metabolic disease (45.6%) topped the list among those ages 50 to 64. Cardiovascular disease was the most common underlying condition for adults ages 65 to 74 and those 75 and up (57% and 69.3%, respectively). Nearly 11% of people hospitalized with the flu last year had no underlying medical conditions. As for vaccine coverage, 32.4% of those admitted had received a flu vaccine. While in the hospital, 84.8% of patients received antiviral therapy, though only 61.6% of children 5 to 17 years were given antivirals. That treatment-rate gap is "suboptimal," the study authors noted, "since all patients hospitalized with influenza should receive prompt antiviral treatment, provided they do not have contraindications." Study limitations included potential underestimation because of clinician-driven flu testing, missing vaccination status among a large proportion of the hospitalized patients (28.5%), and potential issues with generalizing the results to the full U.S. population.
https://www.medpagetoday.com/infectiousdisease/uritheflu/117446
Summary of NACI statement of April 30, 2025
On April 30, 2025, the Public Health Agency of Canada (PHAC) released the National Advisory Committee on Immunization's (NACI) Statement on seasonal influenza vaccines for 2025–2026. This guidance is based on current evidence and NACI expert opinion.
- Seasonal influenza vaccination is the most effective way to protect against influenza and its complications, including hospitalization and death.
- Every fall and winter, influenza and its complications place increased pressure on the healthcare system, especially as other respiratory viruses, such as COVID-19 and respiratory syncytial virus (RSV), circulate. In addition to protecting individuals against influenza and its complications, influenza vaccination helps reduce strain on healthcare resources during these peak seasons.
- Influenza vaccines that meet Health Canada's regulatory requirements for safety, efficacy, and quality are approved for use in Canada. Several types of influenza vaccines are authorized for use, including inactivated, recombinant, and live vaccines, some of which are targeted to specific age groups and health needs.
Following a thorough review of the evidence, NACI makes the following recommendations:
- Influenza vaccine should be offered annually to anyone 6 months of age and older who does not have a contraindication to the vaccine. Patients and providers should also be aware that risks of acquiring influenza are higher in some settings and risks from influenza infection are higher in some individuals than others. Immunization is particularly important for the following groups:
- People at high risk of severe disease, influenza-related complications, or hospitalization;
- All children 6 to 59 months of age;
- Adults and children with certain chronic health conditions;
- All pregnant women and pregnant individuals;
- All individuals of any age who are residents of nursing homes and other chronic care facilities;
- Adults 65 years of age and older; and
- Individuals in or from First Nations, Inuit, or Métis communities.
- People capable of transmitting influenza to those at high risk;
- Health care and other care providers in facilities and community settings;
- Household contacts, both adults and children, of individuals at high risk, whether or not the individual at high risk has been vaccinated;
- Those providing regular childcare to children 0 to 59 months of age, whether in or out of the home; and
- Those who provide services within closed or relatively closed settings to people at high risk (e.g., crew on a cruise ship).
- People who provide essential community services (including health care workers); and
- People whose occupational or recreational activities increase their risk of exposure to avian influenza A viruses (e.g., H5N1).
- People at high risk of severe disease, influenza-related complications, or hospitalization;
- Influenza vaccine should be offered as a priority to the groups for whom influenza vaccination is particularly important (listed above).
- The complete list of individuals at high risk for influenza-related complications or hospitalization, as well as those capable of transmitting influenza to these high-risk groups, can be found in the statement.
For the full statement, including supporting evidence and rationale, please see NACI's Statement on seasonal influenza vaccines for 2025–2026.
Download in PDF format
(256 KB, 6 pages)
COVID-19 & FLU VACCINE
TOP QUESTIONS ANSWERED ABOUT
COVID-19 AND INFLUENZA (FLU) IMMUNIZATION
Will the COVID-19 vaccine protect me against influenza (the flu)?
COVID-19 and influenza are two different viruses that cause two different illnesses. COVID-19 vaccines are designed to best protect you against the COVID-19 virus while the influenza vaccine is designed to best protect you against the flu. The COVID-19 vaccine is not designed to protect you against the flu.
There was hardly any flu last year so why should I get my flu shot this year?
According to experts and based on trends in the past, it is likely that this upcoming flu season might be much stronger and more widespread than the last one. The population’s overall immunity to the flu has declined over the COVID-19 pandemic because of lack of exposure to the latest flu strains. This means that more people might be at risk of getting the influenza virus when it returns. In the past there has been a strong increase in cases after a low outbreak season. Since distancing measures are being lifted, people are returning to offices, schools, public transit, restaurants, etc. This could mean a more widespread influenza outbreak and more severe flu epidemic and put our healthcare systems under significant pressure. In order to protect yourself from influenza, make sure you get your flu shot this year.
Can influenza trigger other non-respiratory illnesses?
Although influenza is primarily considered a respiratory infection, additional evidence suggests that influenza is associated with broader health complications. In fact, influenza can trigger severe cardiovascular complications. In the days following an influenza infection, the risk of heart attack, in otherwise healthy adults, aged 40+, increases more than 10 times while the risk of a stroke increases more than 8 times.
Could I get the COVID vaccine/booster (3rd dose) and the Influenza vaccine this year?
According to CDC and National Advisory Committee on Immunization you can get your COVID-19 vaccine, including the booster (3rd dose) and the flu vaccine at the same time.
If you have any concerns about getting COVID-19 and the flu vaccine at the same time, you should speak with your healthcare provider.
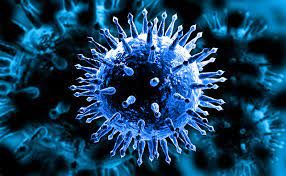
What are the common symptoms for
COVID-19, INFLUENZA, COLD?